File:
<slimemolds.htm> <Index to Mycology> Pooled References <Glossary> Site
Description <Navigate to
Home>
The
Slime Molds: Amoebozoa, Mycetozoa1
(Contact) Please CLICK on underlined
links & included illustrations for details Use Ctrl/F to search
for Subject Matter: Slime molds were considered by
DeBary as a separate group of organisms and he placed them in the
Mycetozoa Bessey included Mycetozoa with
the Protozoa, and Martin placed them in Myxomycetacea with the “True
Fungi.” Here the group will be
treated in the following four classes: Myxogastria: (Myxomycetes;
Mycetozoa), Acrasieae, Plasmodiophoreae (Phytomyxineae) and Labyrinthalae Class: Myxogastria (Myxomycetae)
– True Slime Molds The true slime molds are by far
the largest group in the Amoebozoa, with several hundred species having been
described. These organisms are frequently
encountered on the forest floor, although sometimes they may be found on
lawns, in gardens or other situations.
The vegetative phase is rarely seen by the casual observer because it
develops in a concealed position beneath fallen leaves, underneath bark of
old fallen tree trunks, or even inside the spongy wood mass of a decaying
log. The plasmodium will crawl out to
an exposed and drier situation just before the organism passes into the
fruiting stage. Occasionally such
migrating plasmodia can be found creeping over the outside of a stump or a
log, or over the outside of a pile of dead leaves. However, most often it is the fruiting bodies that are found,
and they bear a resemblance to the fructifications of some true fungi. Two
principal sub-classes of the Myxomycetae are
the Exosporeae
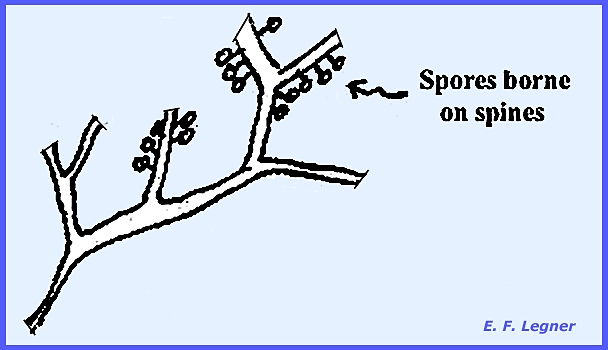
and Endosporae. In the Exosporeae the spores are borne on the outside of the fruiting
body. Ceratiomyxa, a typical
example, has an erect, branched fruiting body on the surface of which are
spines. At the tip of each spine
multinucleate spores without a peridium are formed. In the Endosporeae the spores are borne on the inside of the fruiting
body and a true plasmodium
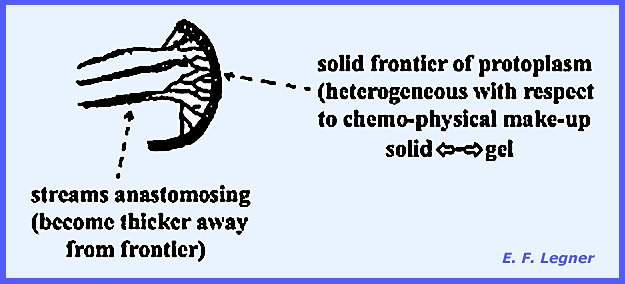
is present. It is an unwalled,
multinucleate mass of protoplasm, capable of ingesting solid food particles
and absorbing water soluble nutrients.
It represents the vegetative stage of the Mycetozoa. This group is
commonly found in cool, shady and moist places. Their shape is irregular and a membrane binds the outer
surface. They are multinucleate with
vacuoles and food granules. A
characteristic streaming action can be seen in these fungi. They may contain various pigments,
and as the plasmodium moves it is capable of absorbing soluble nutrients and
also it can ingest relatively large particles. Wastes are excreted in the path of movement as “ghosts” left in distinctive
patterns. Two plasmodia of similar “race” will fuse and the
protoplasm of one will be engulfed in the other. When incompatible races approach each other, they will not
fuse. No actual contact is necessary
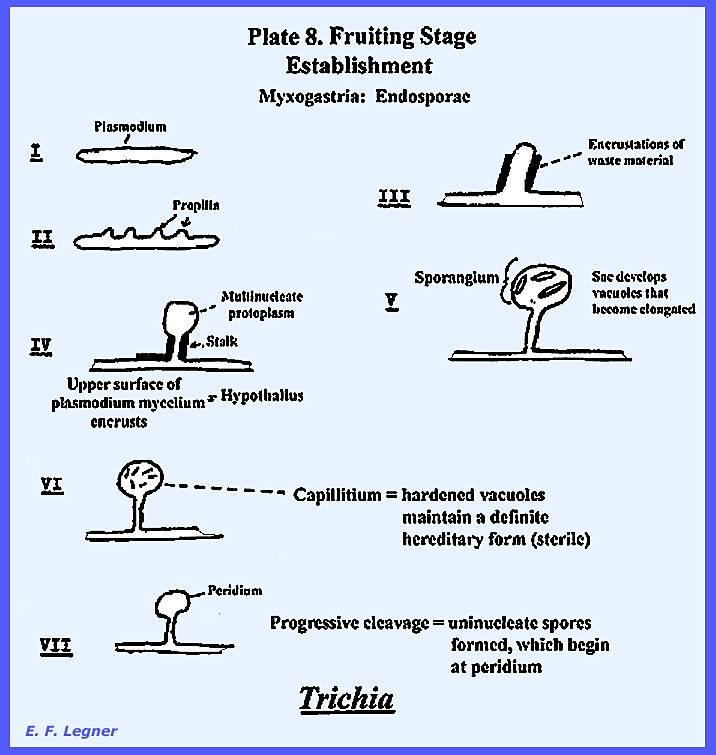
as chemical signals are transmitted across the gap. A fruiting stage is formed when
unsuitable environmental conditions prevail (See PLATE 8 below). Progressive cleavage is where
uninucleate spores are formed beginning at the peridium. Then membranes begin at the peridium and
capillitia. At maturity the peridium
breaks and flakes off and the spores are distributed through the air. Spores may give rise to 1 or 3
protoplasts, which emerge flagellated or non-flagellated. The non-flagellated protoplast, or myxamoebae,
is haploid. The flagellated
protoplast (zoospore,
planospore or
swarm spore)
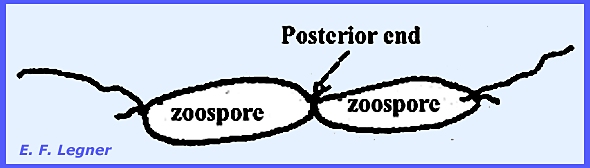
bears two anterior flagella (one short and one long). Two zoospores may fuse at their
posterior ends, which give rise to a zygote (diploid). Several zygotes may fuse to give a
multinucleate diploid zygote. Mitosis
follows. Or, in a rare case one
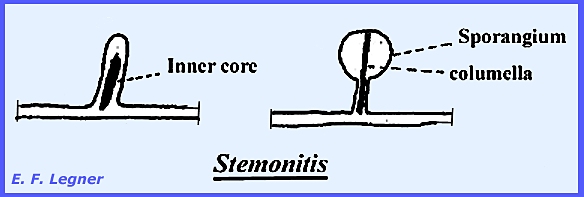
zygote will undergo mitosis to form a multinucleate structure. The inner core may form and ascend through
the sporangium to form a columella. In Stemonitis and Hemitrichia
the vacuoles may anastomoze to form a network capillitium, or, there may no
vacuoles formed. The stalk length may
vary to sessile to form a aethalium (= a fusion of many sporangia), as in Lycogala
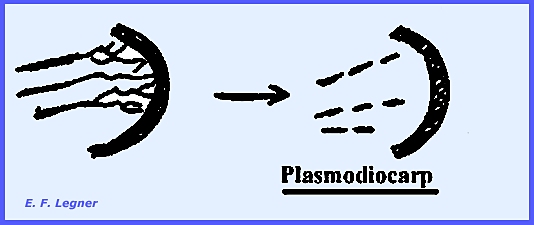
and Fuligo) The inside of the plasmodium may
disrupt to form a plasmodiocarp:
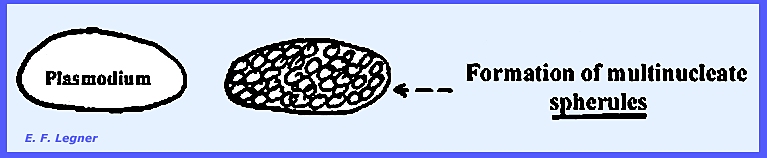
A sclerotium is a mass of multinucleate
cells in the plasmodium called spherules. These are
very resistant to extremes in temperature and desiccation. They may contain 1, 7 or 8 nuclei. When optimum conditions are reestablished,
the walls of the spherules break-up and a plasmodium is reconstituted. Sclerotia will form if the plasmodium is
subjected to slow desiccation, reduced temperatures, or low pH. They are very durable structures, capable
of withstanding the most adverse weather conditions. If formed naturally outdoors they develop
in the location where the plasmodium was growing. ------------------------------------------- Comparison
of Expsporae with Endosporae In the Exosporae the spores are borne on strigmata that cover the outer
surface of the fruiting body. Ceratiomyxa
is the main example of this group. In
the Endosporae spores are
borne within the fructification.
Generally, three morphological types of fruiting bodies are
apparent: (1) small, discrete
sporangia, (2) plasmodiocarps and (3) aethalia. [Please
see PLATE 7 for life cycle of Physarum
polycephalum.] Class: Acrasieae – Cellular Slime Molds
Acrasieae have a characteristic slimy appearance during the vegetative
stage. They are of little economic
importance and the emphasis is rather on their morphogenesis. They are usually isolated from humus soil
where they utilize bacteria. They are
not known to use soluble nutrients and they may be cultured in “pure-mixed”
cultures (growing with a single species of bacteria). The vegetative stage is the myxamoeba, which does not have a
cell wall but rather a cell membrane.
They are uninucleate with food and waste vacuoles. They show a creeping action, engulfing
bacteria by extension of pseudopodia. They
may also exist on dead bacteria and have been grown on the protein fraction
extracted from bacteria. Adverse conditions are passed in a
Microcyst,
which will round up and build a wall around itself. They have the capacity to reduce the density of bacteria in the
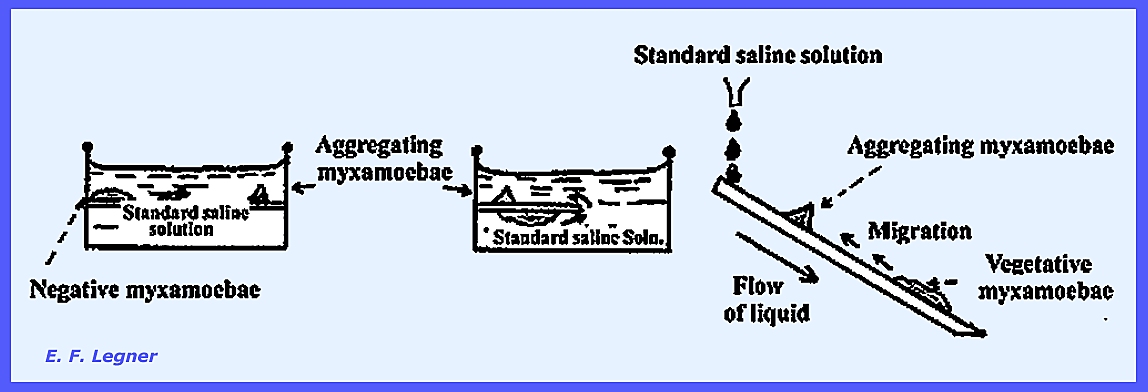
soil. When a minimum density is
reached on a substrate, the myxamoebae will tend to aggregate around certain
centers. These centers are formed by initiator cells,
which secrete Acrasin
that provides a positive chemotrophic attraction to other cells in the
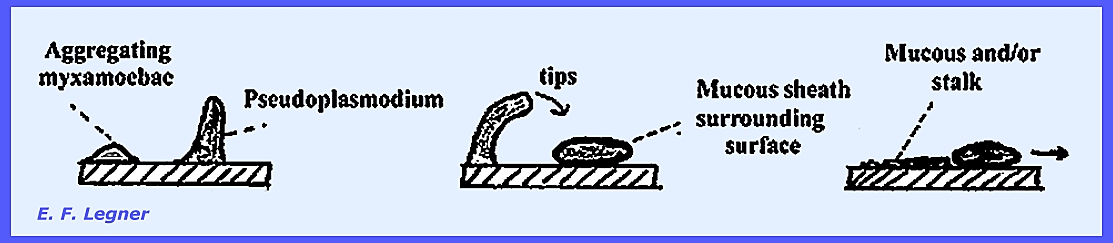
mass. The Fruiting Stage of the Acrasieae
begins with a pseudoplasmodium, which is an aggregation of amoeboid cells
that constitute the initial stage of fruiting. In some species the pseudoplasmodium will not tip. But if it does tip it shows a short
migratory motion. The
pseudoplasmodium then migrates and leaves a mucous sheath behind. Sometimes certain species will leave a
stalk in their trail. While sclerotia
may be interpreted as an interruption in the vegetative development, the formation
of fruiting bodies completes the life cycle of the organism. The spores of most species are very
durable and, similar to sclerotia, they can carry the slime mold over periods
of unfavorable weather. Some species
require a relatively long rest period before they will germinate. During the migratory phase, the
pseudoplasmodium is sensitive to light and temperature, and some feeding
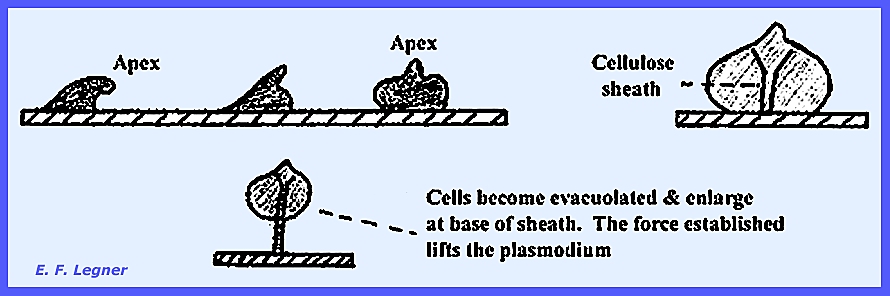
occurs. The Culmination Stage is reached as
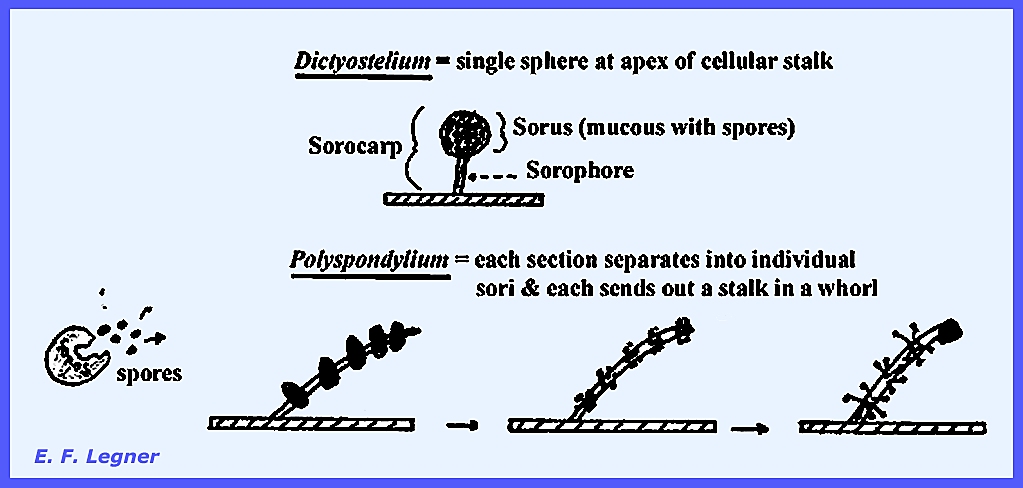
the apex portions of the pseudoplasmodium come to the top. The Sorocarpic Stage varies among
species. In Dictyostelium a
single sphere occurs at the apex of a cellular stalk, while in Polyspondylium
each section separates into individual sori and each sends out a stalk in a
whorl. Comparison
of the Myxogastria and the Acrasieae The Myxomycetae have gametes, a
single-celled plasmodium and flagellated spores. The Acrasieae do not have gametes, a pseudoplasmodium that
gives rise to many cells and no flagellated spores. ------------------------------------------- Also view the following Plates for Example Structures of the
Amoebozoa: Plate 9 = Amoebozoa: Myxogastria, Endosporeae-1 Plate 10 = Amoebozoa: Myxogastria, Endosporeae-2 Plate 11 = Amoebozoa: Myxogastria, Exosporeae and
Amoebozoa: Acrasieae ------------------------------------------- Class: Plasmodiophoreae (Phytomyxineae) The Plasmodiophoreae are the parasitic
slime molds, a group that seemes to be allied to the
Myxomycetes. Some species are
pathogens of economic concern.
Important genera are Plasmodiophora, Spongospora, Sorosphaera
and Sorodiscus All are obligate parasites and intracellular in their
hosts. None form fruiting bodies and
they attack angiosperms primarily.
The genera are grouped by the manner in which spores are held
together. The vegetative phase is an
unwalled amoeboid mass, which is probably diploid and develops within
the cells of the host attached. No
fruiting body is formed, and the entire thallus is ultimately cleaved into
resting spores, which in some cases become firmly joined into a ball (cystosorus)
of definite form. The genera have
been defined on the basis of whether the spores are united or free, and on
the manner of grouping if the spores adhere with one another or are cemented
together. There are over 70 genera with the
life cycle of Plasmodiophora brassicae, the club root of crucifers, being
worked on thoroughly. In this species
the plasmodia reside inside the living cell of the host root. They are picked up as small, multinucleate
plasmodia. These may pass from one
cell to another and although they do not kill the host cell they cause it to
become large and watery (hypertrophy). The
trend is to move toward the cambial layer where they stimulate the host to
divide its cells (hyperplasia). Each daughter cell of the host then
carries over a portion of the plasmodium with it. The plasmodium may fragment into a meront (parent plasmodium = shizont;
fragment = meront). Eventually the
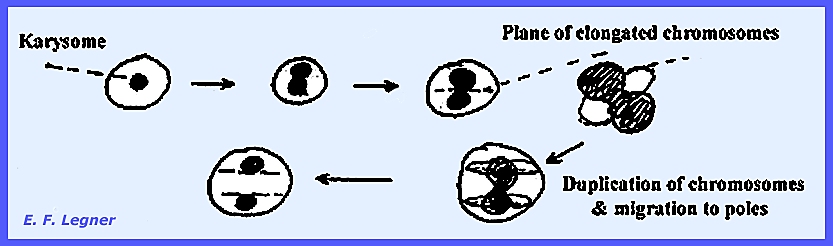
plasmodium will fill the host cell almost entirely. As it enlarges, the nuclei increase in number by protomitosis. At midsummer the nuclei will lose
all karysomes to form the Akaryote State.
The plasmodium becomes vacuolated at this stage that may precede
meiosis. Then the plasmodium splits up
into uninucleate material and finally forms resting spores, which can remain
viable from 7 or 8 years. Under
optimum conditions, the spores break up and release a single anteriorly
flagellated zoospore each. These swim
in free water of the soil until reaching a host. Zoospores penetrate the epidermis of the host, discard their
flagella and become myxamoeba inside the host. The plasmodium that forms becomes multinucleate and is thought
to be haploid. The plasmodium will
break up into parts that form up to 8 nucleate bodies (gametangium). The gametangia will release gametes, each
with two flagella that fuse later on, although there is some doubt as to
whether or not two gametes will fuse from the same gametangium. The fusion of gametes results in a zygote,
which usually takes place near the host epidermis. The zygote divides, becomes uninucleate and migrates to the
host cambium. Plasmodium brassicae is
considered a “High Type” parasite, as it does not readily kill the host
cell. This is in contrast to a “Low Type”
parasite, which either kills the host cell on entering or shortly
thereafter. The nucleus of the host
is the most highly resistant to the pathogen, but it will be greatly
enlarged. Islands of infected cells
surrounded by healthy cells are known as “Krankheitsheid.” The hypertrophy initiated by the pathogen
results in a rather soft host tissue, which is easily attacked by secondary
pathogens. A wilt also results due to
the disruption of the vascular system. Other species and genera differ in
their development from P. brassicae.
In Spongospora subterranean the gametangia may release
zoospores that may infect other hosts.
Here the zoospore-producing bodies are referred to as
“sporangia.” Spongospo. subterranean causes “Powdery Scab”
disease of potato. Sorosphaera
veronicae is a
parasite on Veronica and causes tumors on petioles, stems and leaf
midribs. If there were a sporangium
produced in the order Plasmodiophorales, it would be best to place it with
the Zygomycota. In any case, the
spores tend to be held together in sponge-like masses or cystosori. [In P. brassicae the spores are in no definite mass
pattern].]. In the Genus Sorosphaera
the spores are held together in a sphere; in Tetramyxa they are in
groups of four and in Octomyxa the spores are in groups of eight. ------------------------------------------- Also view the following Plates for Example Structures & Life
Cycles of the Amoebozoa: Plate 6 = Cultures on Agar -- Actinomycetales Plate 7 = Life Cycle -- Physarum polycephalum Plate 8 = Fruiting State -- Myxomycetae,
Endosporae Plate 9 = Example Structures -- Amoebozoa:
Myxogastria, Endosporeae 1 Plate 10 = Example Structures --
Amoebozoa: Myxogastria, Endosporeae
2 Plate 11 = Example Structures --
Amoebozoa: Myxogastria,
Exosporeae and Amoebozoa: Acrasieae
Plate 12 = Life Cycle -- Plasmodiophora
brassicae Plate 13 = Life Cycle -- Spongospora
subterranea Plate 14 = Example Structures -- Amoebozoa:
Plasmodiophoreae Plate
68 = Life Cycle -- Physarum polycephalum Plate
69 = Four types of resting spores: Smooth, Spiny, Reticulate, Warty. Plate
70 = Sporangia types in Myxogastres, Stemonitis,
Dictydium, Physarum & Arcyria. Plate
71 = Three types of capillitium in Myxogastres Plate
72 = Life Cycle -- Plasmodiophora brassicae. Plate
73 = Life Cycle -- Spongospora subterranea. |