File:
<ladybird.htm> [Use Landscape orientation to print] <Navigate to
COMMUNITY INSTRUCTION>
|
<Principal Natural Enemy Groups LADYBIRD BEETLES
Dr. E. F. Legner, University of California, Riverside (Contacts)
Ladybird beetles are part of a large cosmopolitan insect
family, the Coccinellidae, with ca. 252 genera and 3,312 species known as of
1995. They occur in large numbers in
most regions, and are the most often encountered of all predaceous
Coleoptera. Important morphological
characters of these "ladybird beetles" include a short clavate
antenna; head recessed into prothorax; prothorax conspicuously narrower than
elytral bases; tarsal formula 4-4-4, with the 3rd segment reduced; legs short
and stout. The body is usually
subhemispherical, the dorsum highly convex, the venter nearly flat; dorsum
smooth. Their color varies from red
or orange to black Coccinellids are primarily predaceous as larvae and
adults, but some species are phytophagous on green plants, others feeding on
fungal spores. In the subfamily
Epilachninae, mostly in genus Epilachna,
there are several phytophagous species that cause serious injury to legumes,
potato and other crops. Species in
the tribe Psylloborini are fungus-feeders, and one species is
coprophagous. Some species live in
ant nests, and many feed voraciously on aphids, mites, scale insects and
whiteflies and at times on thrips and other insects. Adults usually feed on the same prey
species as the larvae. The
entomophagous species are mainly predaceous on Coccidae, Aphididae and
Aleyrodid ae.Several species of Aiolacaria
and Neoharmonia are effective
predators on all immature stages of some chrysomelids, while other genera and
species favor mites and Chermidae (Clausen 1940/1962). Coccinellidae are important to biological
control, and many species have been successfully imported for the control of
pest insects.
Among aphid and scale
feeding species, there is frequently a pronounced tendency to vary their
diet, so that many will be found at times to feed on immatures of Hemiptera,
Lepidoptera, etc. Some have been
known to feed extensively at nectar glands of plants on sap, pollen, fungi,
honeydew, etc. (Watson & Thompson 1933).
This is especially obvious during times of normal food scarcity and
seems to be a general habit among coccinellids. Chilomenes vicina Muls. feeds extensively on eggs
and young larvae of cotton worm, Prodenia
litura F. in Egypt during times of
aphid scarcity (Bishara 1934). Neocalvia anastomozans Crotch consistently preys on the larvae of
fungus-feeding Psyllobora, also a
coccinellid (Camargo 1937). Both
larvae and adults of Hippodamia tridecimpunctata L. in Japan feed on
eggs and young larvae of the rice beetle, Lema
oryzae Kuway during June and July,
when the preferred aphid hosts are scarce.
This coccinellid is rated as one of the most important natural enemies
of that beetle (Kuwayama cited by Clausen, 1940). The ability to change diet is advantageous because it maintains
the species during host scarcity. A
definite tendency toward cannibalism in both larval and adult coccinellids
serves the same purpose. Schilder
& Schilder (1928) and Balduf (1935) provided early but still valid
information on the food
habits of Coccinellidae.
Cryptolaemus montrouzieri Muls., an Australian
predator of mealybugs, has been effective in reducing heavy infestations in a
number of areas. Because of its size,
it seems not too well adapted to prey on sugarcane mealybugs or other of
similar habit which are protected by leaf sheaths. Cryptognatha noidiceps Mshll., from Trinidad and
tropical America, was responsible for most of the complete control of the
coconut scale, Aspidiotus destructor Sign. in Fiji. An undetermined species closely related to
Cryptognatha, was imported to Cuba
from Malaya in 1930 and was able to control heavy infestations of the citrus
blackfly, Aleurocanthus woglumi Ashby, in just a few
months. Azya trinitatis Mshll.
was the most effective of a series of species introduced for the control of Aspidiotus destructor in Puerto Rico (Clausen 1940/1962). For diaspine scale insect control, coccinellids seem
limited by certain physical characteristics of the scale covering. Species which have been completely or
partially control all had a relatively thin and readily penetrated
covering. Those scales with very
thick and tough overings, such as Chionaspis,
Prontaspis and Lepidosaphes, are relatively free from attack. Coccinellid species that are very
polyphagous among the light scale covering attack group, have been found
unable even to complete development when limited to hosts having a heavy
covering (Clausen 1940). Generally, not much effect has been recorded against
aphids. However, Flint et al. (1995)
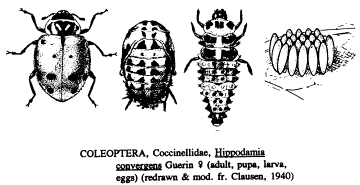
report that releases of the convergent lady beetle, Hippodamia convergens
Guerin-Meneville, which are collected in mountain aggregations in California,
significantly reduced melon aphid numbers on potted chrysanthemum and rose
plants outdoors. Release rates were
quite high, ranging from 34-60 adult lady beetles per potted shrub. This lady beetle is the least expensive
and most widely released aphid predator in the United States. It is unusual because a large portion of
its California population migrates to the mountain ranges when aphid
populations in the Central Valley decrease during summer (Flint et al.
1995). The beetles spend the winter
in massive mountain aggregations, taking in water but consuming no
aphids. They then fly back to the
valley in March when temperatures are warmer. This habit allows collectors to harvest and package large
numbers of the beetles for sale in nurseries and through garden supply
catalogs, etc. Entomophagous Coccinellidae are usually thought of as
being wholly predaceous, but certain species are specialized to the extent
that they may develop as solitary external parasitoids. This is found in some species that attack
hosts much larger than themselves. Novius limbatus Mats., which attacks all stages of the very large Drosicha
corpulenta Kuw. in Japan, is only a
fraction of the size of the adult coccid female. There are times when the egg was laid under the scale and the
resulting larva retained its feeding position on the body venter of a single
host until mature and ready to pupate (Clausen 1940/1962). How effective a
coccinellid is in reducing the host population is related to the relationship
of the larva to its host. The closer
it approaches the habits of a parasitoid the more effective it is in
biological control. Because of this
quality, Rodolia is able to bring
its host to low densities where it is held permanently. The egg is laid on the adult Icerya female or on the egg mass, and
there is enough food material in the egg output of the one female to carry
the larva to maturity. Therefore, the
larva is spared the need to search for food, and the species is able to
maintain itself in an exceedingly low host population density. The same condition operates in species
which are effective against diaspine Coccidae and Aleyrodidae, although in
modified form. These hosts even when
relatively scarce, are gregarious and thus reduce considerably the necessity
of searching for food. The adult
beetle is an active flier and finds the food on which its progeny are to
develop prior to oviposition (Clausen 1940/1962). Aphid-feeding species such as Hippodamia convergens
Guer., which also those which attack solitary Coccidae, often find difficulty
in locating enough hosts in a low population to carry them to maturity. They are often effective in reducing heavy
infestations, but usually only after crop injury has occurred, and their
value is thus reduced. This may be
overcome by spraying the environment with sugar substances that simulate a
high host density (see work by Hagen et al. in section on Manipulation). There are certain specific adaptations in host
relationships that are of interest.
Newly hatched larvae of Cryptognatha
nodiceps under the covering of
mature Aspidiotus scale usually
find a number of eggs which have not be consumed by the parent beetle, and
these provide its first nourishment.
Following emergence from under the scale covering, it feeds mostly on
2nd instar larvae, while following the first molt, attack is extended to any
stage of either sex of the host (Taylor 1935). Young larvae of Scymnus
sieverini Weise feed principally on
young scales of diaspine Coccidae, but the nearly full grown larvae prefer
eggs. Rhizobius ventralis Er.
larvae, which hatch from eggs laid underneath ovipositing Saissetia females may feed either on
the eggs or on the female scale, but those which are free on the foliage attack only young scales (Clausen 1940/1962). Adult coccinellids usually attack the same host species
that serve as food for the larvae, even though a different stage may be
favored. They chew their prey
vigorously and devour all but the harder portions of the body, whereas the
larvae usually bite out a hole in the body wall and suck out the fluid
contents. In some cases a marked
degree of pre-oral digestion occurs, in which the fluid contents are sucked
out and repeatedly pumped back into the prey, thus effecting a rapid and
thorough mixing with the digestive juices (Clausen 1940/1962). The amount of food consumed is proportional to the
predator's size. Clausen (1916)
provided feeding records of a number of California coccinellids, which
indicate that the 4th instar larvae of species of average size, such as H. convergens,
consume ca. 50 aphids per day and that adult females, if ovipositing, have
very nearly the same capacity. The
giant Caria dilatata F. larva of China consumes 400-500 bamboo aphids
daily. Bishara (1934) studying Chilomenes vicina Muls, normally an aphid feeder, found it to destroy up to
22 eggs or 12-15 young larvae of Prodenia
litura F. daily during times of
aphid scarcity. This same rate was
recorded for Coccinella undecimpunctata L. Oviposition.-- The
kind of host insect attack determines the manner and place of
oviposition. Most species that feed on
aphids, such as H. convergens lay their eggs in compact
clusters of 10-50, the spindle-shaped eggs standing vertically on the leaf or
bark surface. However, Synoncha grandis Thbg. spaces the eggs at intervals of several
millimeters. When attacking aphids on
pine and bamboo, Caria dilatata F. places the eggs in two
rows, averaging a total of 28 in each group.
When these are placed on pine needles, a mucilaginous ring is formed
about the needle a few mm. below the mass of eggs (Liu 1933). This is though to provide a degree of
protection from predators.
Coccinellids that feed on red mites and some of the species that
attack diaspine scales lay their eggs singly or in small clusters, and
horizontally, in the vicinity of the hosts.
However, the latter more often place them singly beneath empty scale
coverings, the ovipositor being inserted beneath the margin, through a
feeding hole that was made by the female, or sometimes through a parasitoid
emergence hole. This kind of behavior
is frequent among those species attacking scales that have a soft covering
such as Aspidiotus destructor and related species. Species of genera Chilocorus, Scymnus, Cryptognatha, Pentilia and Rhizobius
usually oviposit in this manner.
Several species that attack Aleyrodidae consistently lay the eggs
singly or in pairs within the pupal cases from which the whiteflies have
emerged. In attacking lecaniine
Coccidae such as Saissetia oleae Bern., that have a large egg
chamber under the female's body, Rhizobius
ventralis and others insert their
eggs under the living host adult. The
mealybug predators usually lay their eggs abundantly over the hosts, directly
on the dorsum of the female scale or in one of the grooves on the surface of
the egg sac (Clausen 1940/1962). Reproduction.-- Reproductive capacity is
usually relatively high, with 1,550 eggs secured by E. K. Carnes (cited by
Clausen, 1940) from a female H. convergens during slightly more than 2
months. Swezey (1905) secured a max.
of 944 from Callineda testudinaria Muls. It may be concluded that the aphid feeding
species of genera Coccinella, Callineda, Leis and Hippodamia lay
the greatest number of eggs, which ranges from 500-1,000. Those that attack diaspine Coccidae,
Aleyrodidae and red mites produce much less.
The oviposition period is quite long, usually exceeding one
month. In some cases it has extended
over 3-4 months, but this is usually associated with lower temperatures and
food scarcity. Oviposition rate is
governed by the same factors, seldom exceeding 10-12 per day over an extended
period even in the most prolific species (Clausen 1940/1962). Mating usually occurs within 1-2 days after emergence, and
fertile eggs are laid 7-10 days later.
Older females that have had sufficient time for egg formation before
mating will produce fertile eggs in a much shorter period of time,
however. Virgin females of several
species have been observed to lay a much smaller total number of eggs than
mated females. However, unfertilized
do not hatch, as they do in Hymenoptera.
In many cases only a single mating is necessary to ensure
fertilization of eggs deposited during the female's entire lifetime (Clausen
1940/1962) Developmental Stages.--Eggs of larger aphid
feeding coccinellids are uniformly spindle-shaped and yellow or
orange-yellow. Species attacking
diaspine Coccidae, Aleyrodidae and red mites have eggs with their poles much
more broadly rounded. They may be
yellow, white or greenish-yellow, with the chorion often bearing minute
reticulate markings. Eggs of Cryptolaemus montrouzieri are amber in color, those of Rodolia cardinalis are
distinctly orange. There is a noticeable darkening of the eggs as they
incubate. Just prior to hatching, the
egg becomes almost blain species that have dark colored larvae, while in
others it becomes grayish. Egg color
is influenced to a considerable extent by the color of the host insects on
which female beetles feed. Larvae of larger aphid feeding
coccinellids, such as Coccinella
and Hippodamia, have variable color
markings and bear a number of relatively short setae on their segments. This is also true of many species that
attack Coccidae. In Chilocorus and related species, the
larvae may bear large, branched fleshy processes on each segment. Others are white, with delicate
setae. Many species of Hyperaspis, Scymnus, Cryptolaemus,
etc. bear a heavy covering of white waxy material, which may be in the form
of granules, slender threads, tufts or plates, depending on the species. These are produced as a glandular
secretion. There seems to be a
tendency among the species attacking mealybugs and other hosts having a waxy
covering to bear a similar covering themselves. This is the result of feeding on hosts with a high wax content
rather than as an adaptation for protection.
However, some species developing on diaspine Coccidae have this heavy
waxy covering while others on the same host do not (Clausen 1940/1962). Early work on the morphology and
classification of coccinellid larvae may be found in Böving (1917) and Gage
(1920). Coccinellids usually have 4 larval instars, with
exceptions being Pseudonycha japonica Kuris, which Iwata (1932)
found to have 5, and Hyperaspis lateralis Muls. in which the autumn
generation has only 3 larval instars contrasted to the normal 4 of the spring
generation (McKenzie 1932). They usually pupate in situ
on the foliage or bark at the point where they had fed. However, Cryptolaemus montrouzieri
frequently descend the tree trunk and pupates in masses in sheltered places
thereon or in trash on the ground surface.
Chilocorus similis and Chilocorus spp. and Cryptognatha
assemble for pupation in large aggregations on the twigs, the lower sides of
main branches and the trunk (Clausen 1940/1962). When ready to pupate, the mature larva fastens the caudal tip
of the body securely to the substrate by means of a mucilaginous
secretion. Aphid-feeding species
generally cast the final larval exuviae almost completely, and it remains only
as a collar or ring about the abdomen base.
Rodolia, Cryptolaemus and some species of Curinus and Scymnus just effect a median split of the exuviae over the
anterior body portion (Clausen 1940/1962). Some Examples of Ladybird Beetles
Key
References: Clausen, C. P. 1962. Entomophagous Insects. Hafner Publ., Co., NY. 688
p. Flint, M. L., S. H. Dreistadt, J. Rentner
& M. P. Parrella. 1995. Lady beetle release controls aphids on
potted plants. Calif. Agric. 49(2):
5-8. Pacheco, F.
1986. Plagas de los Cultaivos Agrícolas en Sonora y Baja
California. Sec. Agr. Recursos
Hidra., Ins. Nac. Invest. Agr., Cent. Invest.
Agr. Noroeste, Campo Agr. Exptal. Valle del Yaqui, Cd. Obregón, Son.,
México. 414 p. |